Direct-to-consumer (DTC) microbiome testing kits have become increasingly popular, promising consumers personalized insights into gut health based on a simple stool sample. But how reliable are these tests?
A new study published in Communications Biology by a team of researchers from the National Institute of Standards and Technology (NIST) and the Center for Advanced Microbiome Research and Innovation (CAMRI) at the University of Maryland School of Medicine suggests that results can vary widely depending on which company performs the analysis.
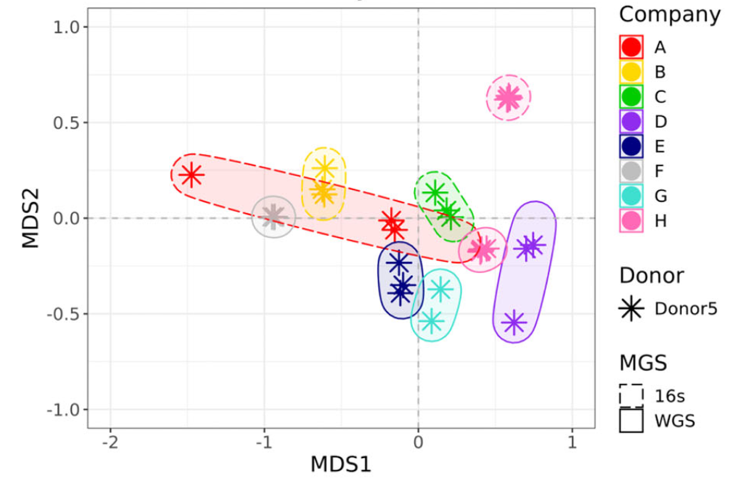
The study evaluated the analytical performance of seven commercial gut microbiome testing services, using a standardized human fecal reference material developed by NIST. By submitting identical samples to multiple providers, the researchers were able to directly compare the taxonomic profiles and interpretations generated by each company.
Wide variation in reported microbiomes
The results revealed substantial discrepancies between testing services. Different companies reported very different microbial compositions, even though they analyzed the same biological sample.
In fact, the variation between companies was comparable in magnitude to the biological variation typically observed between different individuals, raising concerns about the reliability and reproducibility of current DTC microbiome testing services.
The researchers attribute much of this variability to methodological differences, including variations in DNA extraction, sequencing approaches, bioinformatics pipelines, and thresholds for reporting microbial taxa. Many companies also provide limited information about their analytical methods, making it difficult to compare results across platforms.
Implications for consumer health
The global microbiome testing market has expanded rapidly in recent years as public interest in the microbiome and personalized health has grown. Many companies now offer microbiome reports that include dietary recommendations, lifestyle advice, or health assessments based on the detected microbial profile.
However, the new study highlights a key issue: analytical validity must come before clinical interpretation. Without reliable, reproducible measurements of microbiome composition, recommendations based on these tests may be misleading or unsupported by evidence.
The authors emphasize that consumers may not realize that these services occupy a regulatory gray areabetween strictly regulated medical diagnostics and loosely regulated wellness products.
The need for standards in microbiome testing
To address these challenges, the study calls for the development of clear standards and quality-control frameworks for microbiome testing services. Standardized reference materials, such as those developed by NIST, could help laboratories benchmark their analytical pipelines and improve comparability across platforms.
Establishing such standards will be essential as microbiome science moves toward clinical applications, including diagnostics and microbiome-based therapeutics.
Advancing responsible microbiome innovation
The research reflects CAMRI’s broader commitment to ensuring that microbiome science advances with strong methodological rigor and transparency.
“Microbiome research holds tremendous promise for improving human health,” the authors note. “But realizing that promise requires robust analytical methods and clear standards that ensure data accuracy and consumer confidence.”
As interest in personal microbiome testing continues to grow, studies like this one provide an important reminder that scientific validation and quality control must keep pace with innovation.
The study has received extensive media coverage, available here.
Reference: Servetas, S. L., Gierz, K. S., Hoffmann, D., Ravel, J. & Jackson, S. A. Evaluating the analytical performance of direct-to-consumer gut microbiome testing services. Commun. Biol. 9, 269 (2026).
March 14, 2026